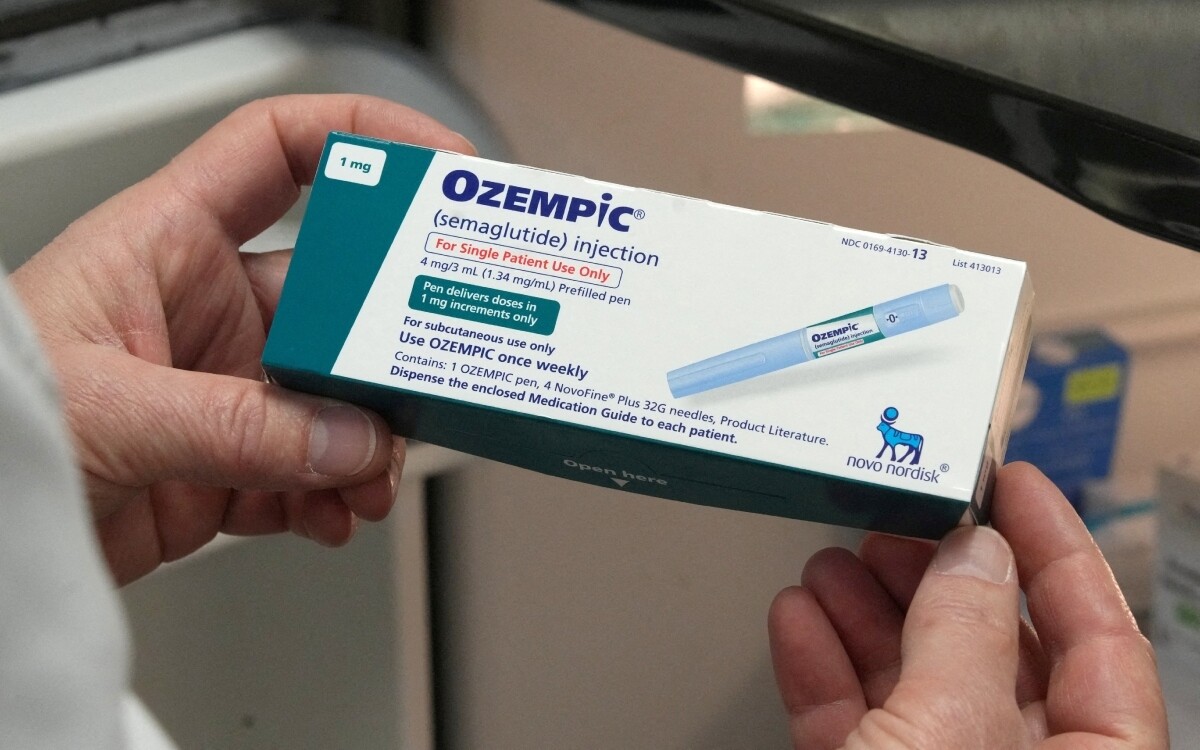
The Federal Commission for the Protection against Sanitary Risks (COFEPRIS) has issued a health alert after detecting counterfeit Ozempic 1 mg (semaglutide) pre-filled pens. The agency warns that this batch presents multiple anomalies, making it dangerous for consumers. According to the technical-documental analysis, the counterfeit product does not contain the active ingredient, meaning it has no semaglutide, the component that provides its therapeutic effect. COFEPRIS states that the counterfeit pens lack labeling, the texts and legends are in a language other than Spanish, and the batch numbers do not match between the secondary packaging and the pre-filled pen. The agency notes that this "represents a risk to the health of the population, since its origin, manufacturing, storage, distribution, and transport conditions, as well as the raw materials used in its preparation, are unknown." COFEPRIS reminds the public that Ozempic is a prescription medication and its indiscriminate use without medical supervision can be a risk. The authority has issued the following recommendations: inspect the primary and secondary packaging visually before use, verify that batch numbers and expiration dates match, and that the pen shows no signs of tampering. Do not purchase medications from e-commerce platforms, websites, mobile apps, the street, tianguis (fairs), or informal establishments, especially those that require a prescription, are offered at prices well below the market, or are in a language other than Spanish. If you have used this product, stop its use immediately and consult a healthcare professional. If you identify a product with the described anomalies, do not purchase or use it. COFEPRIS asks the population to report any information on the commercialization of this counterfeit product through the health complaints portal.















